Fluid and Electrolyte Basics
Osmosis — passive transport of water across a semipermeable membrane. Water tends to move across the membrane until the solute concentration (the amount of solids) on both sides are the same. When water concentration is high in one compartment, there will be a net diffusion of water into the other compartment and also a diffusion of solutes to equalize the osmolarity in the two compartments. The volumes will not change.
Osmotic Pressure — the tendency for water to diffuse to higher solute concentration can be measured as a pressure:
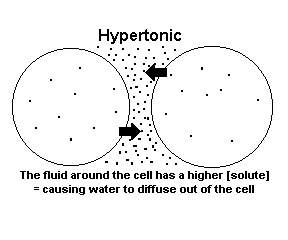
a) Hypertonic Solution — the solution around the cell has a higher [solute] (called osmolarity) than the cytoplasm ==> water diffuses out of the cell and the volume of the cytoplasm decreases affects both cells with and without cell walls. cells lose water and shrink
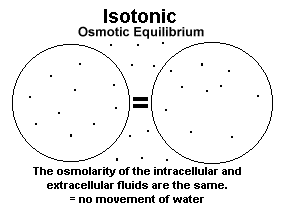
b) Isotonic Solution — solution around the cell has the same osmolarity as the cytoplasm ==> no net movement of water. At osmotic equilibrium, the osmolarity of the intracellular and extracellular fluids are the same. A fluid into which a cell can be placed without causing either swelling or shrinkage is said to be ISOTONIC with the cells. Iso-ionic refers to a similar concentration of a particular electrolyte in a solution compared to extracellular fluid; therefore solutions which are iso-ionic with regard to sodium will have sodium concentrations comparable to the sodium concentration of extracellular fluid and plasma.
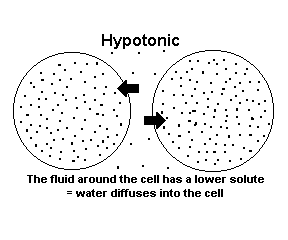
c) Hypotonic Solution — the solution around the cell has a lower osmolarity than the cytoplasm ==> water diffuses into the cell; water moves into the cells and they swell
Distribution of Fluids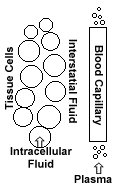
Water in the body is in a constant state of motion, shifting between the three major fluid compartments of the body. These are:
1. Blood plasma (intravascular volume) (the fluid outside cells or extracellular fluid)
2. Interstitial fluid (the fluid outside cells or extracellular fluid)
3. Intracellular fluid (the fluid inside cells)
Normal Body Water Distribution
Dehydration
The majority (probably >90%) of dehydration cases have sustained isotonic losses of fluids. Gastrointestinal and urinary losses of fluids result in the loss of water AND electrolytes including sodium, potassium, chloride and bicarbonate. The amount and type of electrolytes lost depend on the disease that is causing the abnormal loss. The fluid and electrolytes which are lost originate initially from the extracellular (plasma water + interstitial water – fluids outside of cells) compartment.
Approximately 90% of the body’s water intake comes via the gastro-intestinal tract. The remaining 10% is called metabolic water and is produced as the result of various chemical reactions in the cells of the body’s tissues.
Once an ingested meal enters the GI tract, fluids shift from the plasma, making the contents isotonic. Water movement into and out of the GI tract follows the osmotic gradients created by electrolyte movement. For this reason, providing solid food to a dehydrated animal is contradicted, and could prove fatal.
| Rehydration | |
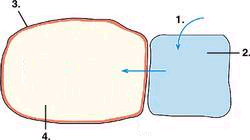 |
1. excess fluids are added to extracellular fluid compartments 2. (interstatial fluid and plasma) 3. Cell membrane 4. and water moves to intercellular compartment by osmosis |
| Dehydration | |
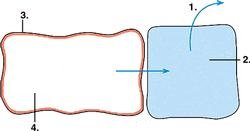 |
1. water loss from extracellular fluid compartments 2. (interstatial fluid and plasma) 3. Cell membrane 4. water leaves intercellular fluid compartment by osmosis |
Estimating the percentage dehydration based upon physical examination findings.
|
Estimated Percentage Dehydration |
Physical Examination Findings |
|---|---|
|
<5 |
History of fluid loss but no findings on physical examination |
|
5 |
Dry oral mucous membranes but no panting or pathological tachycardia; slight loss of skin elasticity |
|
7 |
Mild to moderate decreased skin turgor, dry oral mucous membranes, slight tachycardia, and normal pulse pressure; definite loss of skin elasticity; slight prolongation of capillary refill; slight sinking of eyes into orbit; slight dryness of oral mucous membranes. |
|
10 |
Moderate to marked degree of decreased skin turgor, dry oral mucous membranes, tachycardia, and decreased pulse pressure. prolonged capillary refill; eyes sunken in orbits; dry mucous membranes; possible signs of shock |
|
12 |
Marked loss of skin turgor, dry oral mucous membranes, and significant signs of shock. |
Fluid Replacement
When designing a fluid therapy plan one needs to decide for each of the three constituents: How much fluid to use, Route of administration, Which replacement fluid to use.
| How much fluid to use | ||
|---|---|---|
| 135 X Weight (in kgs) = Maintenance Requirements per day |
1. Determine the maintenance requirement (replacement of normal fluid losses via urine, feces, respiration, skin). Requirements are usually 40-60 ml/kg/day but very young animals require 135 ml/kg/day. | |
| Maintenance X Percentage of Deficit | 2. Establish the percentage of dehydration using the table and multiply the percentage of dehydration by the maintenance requirement to determine the deficit amount. | |
| Deficit X .75 | 3. Provide 75% of the deficit amount during the first 24 hours. | |
| 4. Must also add “daily maintenance volumes” to your calculated deficit volume if the animal is not eating nor drinking. | ||
| Route to administer | ||
| PO: | Oral route; The absorption of fluids, electrolytes and foods from the stomach is slow. Obviously the oral route of delivery of fluids is contraindicated if the animal needs rapid fluid loading, such as in states of shock. Hypertonic solution, such as 50% dextrose should not be given orally, as the osmotic draw of these hypertonic solutions will bring water into the stomach, causing gastric distension. (concentrations of 20-50% are used to treat hypoglycemia). Gastric distension is one stimulus for vomiting. Whenever the oral route of administration is used, there is always the danger of aspiration of fluid and food into the airways. | |
| Sub-Cutaneouly: | (hypodermoclysis); The administration of fluids subcutaneously is a rapid, easy method of providing fluids. The absorption of fluids from the subcutaneous space is relatively slow, occurring over 6-8 hours. If the animal is severely dehydrated, blood is shunted away from the subcutaneous tissues to more vital structures, and absorption of fluids will be delayed even longer. Place about 10-20 ml/kg/hour per site. | |
| IV: | large fluid volumes can be administered rapidly; 10 ml/kg/hour; can only be administered by lisenced professional. | |
| Which fluid to use | ||
| Select a replacement fluid that closely matches the osmolality of the extracellular fluid (plasma) to acheive the highest rate of safe absorption. The osmolality of blood plasma sets the standard. Fluids in the range of 240 to 340 mOsm/liter are considered isotonic. Fluids with tonicities above 340 mOsm/liter are hypertonic; those with tonicities below 240 mOsm/ liter are hypotonic. | ||
|
Buffer |
Calories |
Osmolality |
|||||||
|
Solution |
Na |
K |
Cl |
Ca |
Mg |
mEq/L |
Kcal/L |
mOsm/L |
|
| Plasma |
144 |
5 |
107 |
5 |
1.5 |
– |
– |
290 |
|
| Dextrose 5% in Water |
– |
– |
– |
– |
– |
– |
170 |
278 |
|
| Dextrose 2.5% in 0.45% Saline |
77 |
– |
77 |
– |
– |
– |
85 |
280 |
|
| Ringer’s Lactate Solution |
130 |
4 |
109 |
3 |
– |
Lactate 28 |
9 |
272 |
|
| Ringer’s Solution |
147 |
4 |
156 |
4.5 |
– |
– |
– |
309 |
|
| Normosol-R (Multisol-R) |
140 |
5 |
109 |
– |
3 |
Acetate 27 Gluconate 23 |
15 |
294 |
|
| Dextrose 5% in Ringer’s Lactate |
130 |
4 |
109 |
3 |
– |
Lactate 28 |
179 |
525 |
|
| Normal Saline (0.9%) |
154 |
– |
154 |
– |
– |
– |
– |
308 |
|
| Dextrose 50% |
– |
– |
– |
– |
– |
– |
1700 |
2525 |
|
| Dextrose 5% in Saline (0.9%) |
154 |
– |
154 |
– |
– |
– |
170 |
– |
|
| Ringer’s, Lactated Ringer’s (LRS) is isotonic and closely matches the extracellular fluid composition; 0.9% NaCl (normal saline) is an isotonic solution of Na, Cl, and water; 5% dextrose is an isotonic solution of dextrose in water; the dextrose is rapidly metabolized, resulting in the administration of free water
References and adaptations from: |
|||||||||